In short yes, but it depends which peptides, for what use, and how “work” is defined. Peptides are a broad class of molecules (short chains of amino acids) that include established prescription drugs, promising new therapeutics, cosmetic ingredients, and over-the-counter “performance” supplements. The strength of evidence ranges from rigorous randomized trials (strong) to small studies, mixed results, or mechanistic claims without clinical proof (weak). Below are five unbiased theses that lay out the evidence, limits, and practical takeaways.
Therapeutic peptides (prescription drugs) have clear, high-quality evidence for many #SSWdiseases.
Peptide drugs, examples include insulin analogues, GLP-1 receptor agonists (semaglutide), and newer dual/triple agonists (tirzepatide, etc.), have repeatedly shown clinically meaningful benefits in large randomized controlled trials and are approved by regulators. For example, once-weekly semaglutide produced sustained, clinically relevant weight loss in phase 3 trials, and newer multi-agonists show even larger effects in recent high-quality trials. Reviews of peptide therapeutics describe a growing number of approved peptide drugs and robust clinical momentum in the field. PubMed+2New England Journal of Medicine+2
For prescription indications (diabetes, obesity, certain endocrine and metabolic disorders, some rare diseases), peptides do work, the evidence is high quality and regulatory approvals back them.
Topical and cosmetic peptides show promise, but clinical evidence is mixed and often limited by small studies or formulation issues.
Many skincare products contain peptides (signal peptides, carrier peptides) intended to stimulate collagen, modulate inflammation, or reduce wrinkles. Some mechanistic and small clinical studies show improvements in skin hydration, elasticity, or biomarkers; systematic reviews for oral hydrolyzed collagen report modest improvements in skin hydration/elasticity. However, translating peptide activity into effective over-the-counter topical creams depends on formulation, dose, skin penetration, and independent replication, areas where the evidence is more heterogeneous than for prescription peptide drugs. PMC+1
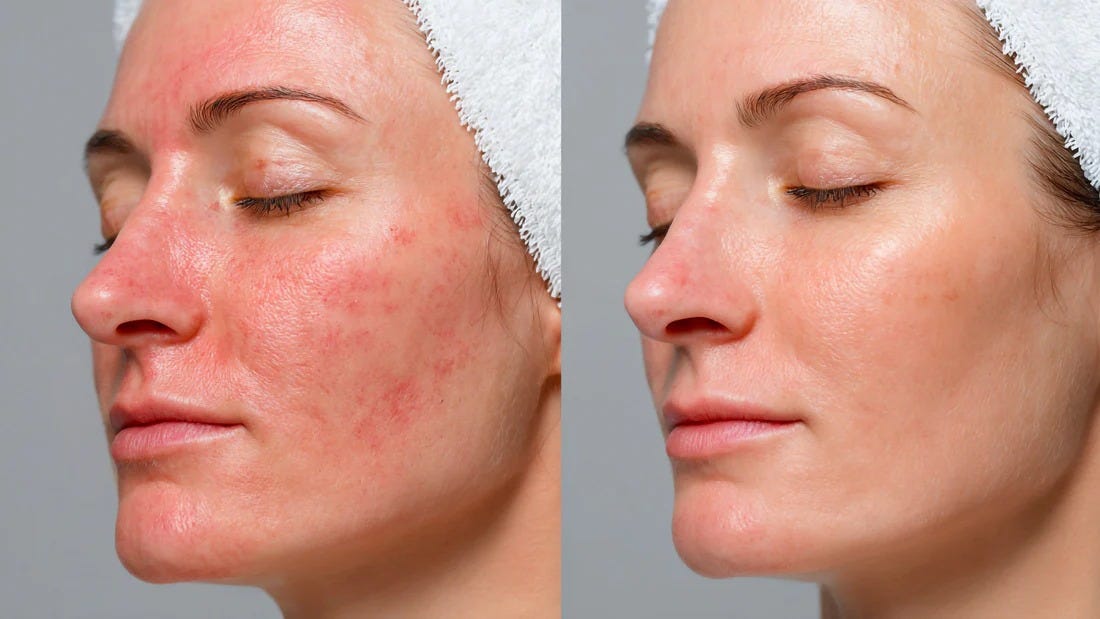
Results after using the famous GHK-Cu copper peptide for skin care.
Cosmetic peptides may help in some products/settings, but claims vary widely; look for peer-reviewed RCTs on the specific product or ingredient rather than trusting marketing alone.
“Performance” and hormone-modulating peptide supplements (e.g., growth-hormone secretagogues, amino-acid mixes) have limited, inconsistent clinical support and safety/regulation concerns.
A number of over-the-counter peptides and amino-acid formulas claim to boost growth hormone, muscle mass, or recovery. Some small studies (or physiologic studies showing transient hormone changes) exist, but high-quality, long-term trials demonstrating meaningful, safe gains in strength, clinically relevant muscle mass, or athletic performance are sparse. Importantly, many of these products are poorly regulated, may be mislabeled, or carry safety risks if used incorrectly. Meta-analyses of growth-hormone therapy show specific effects in medical settings, but that does not validate unregulated supplement use. PMC+1
Be skeptical of supplement claims. Medical hormone therapies have evidence but require supervision; unregulated peptides/supplements often lack robust efficacy and may pose safety or legality questions.
Peptides’ advantages (target specificity, low immunogenicity) are real but are balanced by practical challenges: delivery, stability, and manufacturing quality.
Peptides often bind targets with high specificity and can be chemically modified to improve half-life or tissue targeting. Yet they can be broken down by enzymes, may need injections rather than oral dosing, and require careful synthesis and quality control. Regulatory agencies (like the FDA) publish guidance for peptide drug development and increasingly approve peptide/oligonucleotide medicines as the field matures—evidence that peptides are a viable drug class but not trivially interchangeable with small molecules or supplements. Nature+1
The biological advantages of peptides are genuine, but successful clinical use depends on rigorous drug development (formulation, delivery, manufacturing) and regulatory oversight.
Safety and regulation matter evidence and approvals should guide use; individual claims require scrutiny.
Where peptides are prescription medicines supported by phase-3 trials and regulatory approval, benefit–risk profiles are characterized and clinicians can manage side effects (for example, gastrointestinal effects with GLP-1 agonists). For cosmetics and supplements, regulatory oversight is weaker and evidence is variable. Consumers and clinicians should rely on peer-reviewed trials, regulatory approvals, and independent safety data rather than marketing or anecdotes. The FDA and other agencies provide guidances and track peptide approvals, reflecting both opportunity and necessary caution. U.S. Food and Drug Administration+1
Use prescription peptides under medical supervision; treat supplements and cosmetics skeptically until high-quality evidence exists.
Conclusion
Peptides do work in many contexts; they are an established and expanding class of prescription medicines with strong clinical evidence for several major indications (diabetes, obesity, endocrine disorders, some rare diseases). For cosmetics and dietary supplements the evidence is mixed and product-specific. For performance/hormone claims, evidence is weak or inconsistent and safety/regulatory issues are important. In short: trust the evidence and the regulatory status of the particular peptide you’re asking about.
Article written by George Vrabec
Selected sources & further reading
(links below provide the underlying evidence and reviews cited above)
Wang L., et al., Therapeutic peptides: current applications and future directions — review summarizing peptide drug development and applications. Nature
Wilding J.P.H., et al., Once-Weekly Semaglutide in Adults with Overweight or Obesity (NEJM, 2021) — pivotal RCT showing clinically meaningful weight loss with a peptide drug. PubMed
NEJM (2025) Tirzepatide vs Semaglutide — large recent trial showing substantial weight loss with newer multi-agonist peptides. New England Journal of Medicine
Pu S.Y., et al., Effects of Oral Collagen for Skin Anti-Aging: A Systematic Review and Meta-Analysis (2023) — meta-analysis suggesting modest benefits of oral hydrolyzed collagen on skin metrics. PMC
U.S. Food & Drug Administration — Guidance for Industry: Synthetic Peptides and clinical pharmacology considerations for peptide drug products — guidance documents describing regulatory standards. U.S. Food and Drug Administration+1
Leave a comment